The Seyferth–Gilbert homologation is a chemical reaction of an aryl ketone 1 (or aldehyde) with dimethyl (diazomethyl)phosphonate 2 and potassium tert-butoxide to give substituted alkynes 3.[1][2] Dimethyl (diazomethyl)phosphonate 2 is often called the Seyferth–Gilbert reagent.[3]

This reaction is called a homologation because the product has exactly one additional carbon more than the starting material.
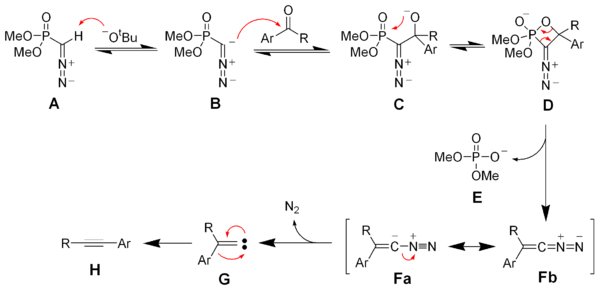
Deprotonation of the Seyferth–Gilbert reagent A gives an anion B, which reacts with the ketone to form the oxaphosphetane D. Elimination of dimethylphosphate E gives the vinyl diazo-intermediate Fa and Fb. The generation of nitrogen gas gives a vinyl carbene G, which via a 1,2-migration forms the desired alkyne H.
The dimethyl (diazomethyl)phosphonate carbanion can be generated in situ from dimethyl-1-diazo-2-oxopropylphosphonate (also called the Ohira-Bestmann reagent) by reaction with methanol and potassium carbonate as the base by cleavage of the acetyl group as methyl acetate. Reaction of Bestmann's reagent with aldehydes gives terminal alkynes often in very high yield and fewer steps than the Corey–Fuchs reaction.[4][5]

The use of the milder potassium carbonate makes this procedure much more compatible with a wide variety of functional groups.
Improved in situ generation of the Ohira-Bestmann reagent
Recently a safer and more scalable approach has been developed for the synthesis of alkynes from aldehydes. This protocol takes advantage of a stable sulfonyl azide, rather than tosyl azide, for the in situ generation of the Ohira−Bestmann reagent.[6]
Another modification for less reactive aldehydes is made by replacement of potassium carbonate with caesium carbonate in MeOH and results in a drastic[quantify] yield increase.[7]
Seyferth-Gilbert反應被Vandewalle小組用於維生素D3衍生物的合成(式 3)。在埃坡黴素B和D及其類似物的合成中,該方法也被用於構築重要片段,收率達 80% (式 4)。

Ohira在1989年報道了一種改進方法。該法以1-重氮-2-氧代-丙基磷酸二甲酯(2)為試劑,在氬氣氛下現場去除酰基生成Seyferth試劑(1)的碳負離子。隨後,Bestmann係統地研究了這一“一瓶”方法,發現該法隻需簡單地通過室溫下把磷酸酯加入K2CO3與醛的甲醇溶液就可合成炔,從而避免使用強堿、低溫和惰性氣體等條件。在這一條件下,脂肪醛和芳醛均可順利實現增碳,收率為72%~97%(式 5)。與α,β-不飽和醛反應雖然也可實現增碳,但產物為甲氧基共軛加成產物4(式 6)。






