Hofmann 降解反應是將伯酰胺通過氧化降解形成少一個碳原子的伯胺的過程。其反應的機理如下:
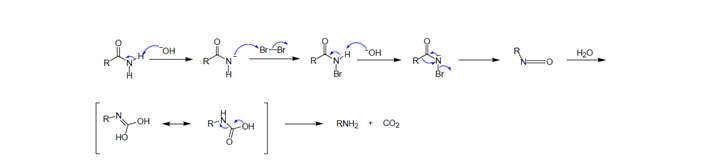
早期的Hofmann降解通常采用NaOH水溶液和液溴來實現,其反應的條件比較苛刻,後來人們對反應的條件進行了不斷地改進,能夠采用不同氧化劑和堿溫和地實現。目前,通常采用的條件:
1,Br-NaOH體係Hofmann降解
2,NaClO做氧化劑的Hofmann降解
3,NBS與DBU體係的Hofmann降解
上麵的幾種體係大家在反應中經常用到,通常會很順利地得到產品。但是在最近所負責的項目中,采用上述條件遇到了一些問題,均沒有直接拿到目標產物,主要由其化合物結構所造成的。底物酰胺連在季碳原子上而季碳原子上又連有芳環或芳雜環結構,因此這種酰胺具有較大的空間位阻,決定了其性質的特殊性。
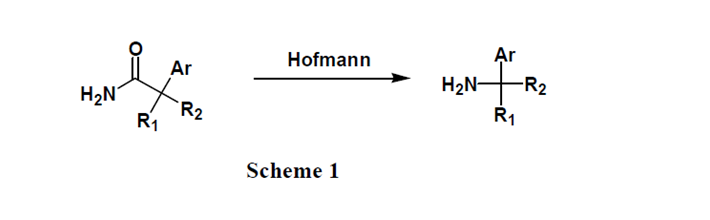
如Scheme 1所示:胺是我們想要得到的目標產物,我們嚐試了三種不同的常用條件:
1,Br2-NaOH 體係Hofmann降解:
A solution of NaOH (5 eq) in water was cooled to 0oC, and treated withbromine (1 eq). After 10 min, the material (1 eq) in acetone was added and themixture was heated to 70-75 oC and stirred for another 45min, the mixture wasdiluted with ethyl acetate, washed with water, brine. TLC showed the reactiongot messy, and no desired product was detected by LC_MS。
2,NaClO做氧化劑的Hofmann降解:
To a solution of NaClO (5 eq) was added the material (1 eq), and thesolution was stirred for 2h at room temperature. TLC showed no reaction, andthe temperature was warmed up to 60-70 oC, stirring for 30min, TLC showed thereaction got messy.
3,NBS 與DBU體係的Hofmann降解:
To a solution of material (1 eq) wasadded the NBS (1 eq), and DBU (1.1 eq) in the methanol. The mixture was stirredfor 20 min at 80 oC, at which point more NBS (1 eq) was added to the mixture,stirred for another 10 min. TLC showed got messy, and no desired product wasdetected by LC_MS.
上麵的三種方法我們都沒有得到目標產物,於是我們參考相關文獻,使用PhI(OCOCF3)2 做氧化劑進行反應,同樣目標產物並沒有得到,但是得到了比較幹淨的新化合物,進過鑒定分析,新的化合物為異氰酸酯中間產物,如下圖所示:

此結構的異氰酸酯能夠穩定地存在,我們采用不同的酸堿條件均可以把中間態異氰酸酯很好地轉化為目標產物胺。具體方法如下:
To a solution of 1 (1eq) was added the PhI(OCOCF3)2 (1.2 eq ) in MeCN at room temperature. The mixture was stirred at ambient temperature for 4hs, the solvent was removed and water added, extracted with ethyl acetate, evaporation, after giving the product 2.
A mixture of potassium hydroxide (3 eq) and the product 2 (1 eq) in tetrahydrofuran: water (1:1) was stirred at room temperature for 12hs. The amine was extracted with ethyl acetate. The organic phase was washed with brine, dried over Na2SO4, and concentrated in vacuo to afford the desired product.
The solution of the product 2 and HCl aqueous in tetrahydrofuran was stirred at ambient temperature for 30min, then extracted with ethyl acetate. The organic phase was washed with brine, dried over Na2SO4, and concentrated in vacuo to afford the desired product.
討論部分:
討論部分:
1.上麵提到的三種方法對於我們做伯,仲碳原子相連酰胺的Hofmann降解反應,通常大都能夠得到很好的結果。
2.采用PhI(OCOCF3)2作為氧化劑,同樣也適用於伯,仲碳原子相連酰胺Hofmann降解反應,相比之下其條件更加溫和,收率也更高。
3.PhI(OCOCF3)2作為氧化劑,在製備季碳原子相連的胺的時候,由於空間位阻大,對有些反應很難進行,或者不能一步得到相應的胺,而是得到能夠穩定存在的中間態異氰酸酯。
4.對於得到的能夠穩定存在的中間態異氰酸酯,可以通過酸堿條件處理得到相應的胺。




